Medicare Drug Price Negotiation: 2025 Highlights & How Pharma Companies Can Prepare for 2026

In 2025, the US prescription drug market faced a turning point when Medicare drug pricing reform took effect. This outcome resulted from long-term discussions on prescription drug prices in the US, which have been largely shaped by complex market dynamics. This also highlighted the ongoing need to improve affordability and access for patients.
The Inflation Reduction Act of 2022 aimed to address this challenge by granting the CMS the authority to negotiate prices for certain high-cost drugs directly. In 2025, CMS completed its first round of negotiations for 10 widely prescribed drugs, with the new prices set to take effect in 2026. A second wave is already in motion, covering 15 additional drugs, and this is where manufacturers need to act now. Negotiations for this next list are happening in 2026, with the final prices going live in 2027.
This means that the companies newly listed in this second wave are now on the clock: preparation for negotiations with CMS must start now.
What is Medicare Drug Price Negotiation?
Medicare drug price negotiations aim to make high-cost medicines more affordable by allowing CMS, under the Inflation Reduction Act, to work directly with manufacturers to set a “maximum fair price” for drugs that account for significant Medicare spending and lack cheaper alternatives.
Replacing the old noninterference rule, the structured process considers clinical benefits, alternatives, R&D costs, and usage data, with input from both sides. This approach helps lower patient costs, supports Medicare’s financial sustainability, and gives manufacturers greater pricing predictability.
Medicare Drug Price Negotiation: What happened in 2025
Some of the most noteworthy changes brought about by the reform are:
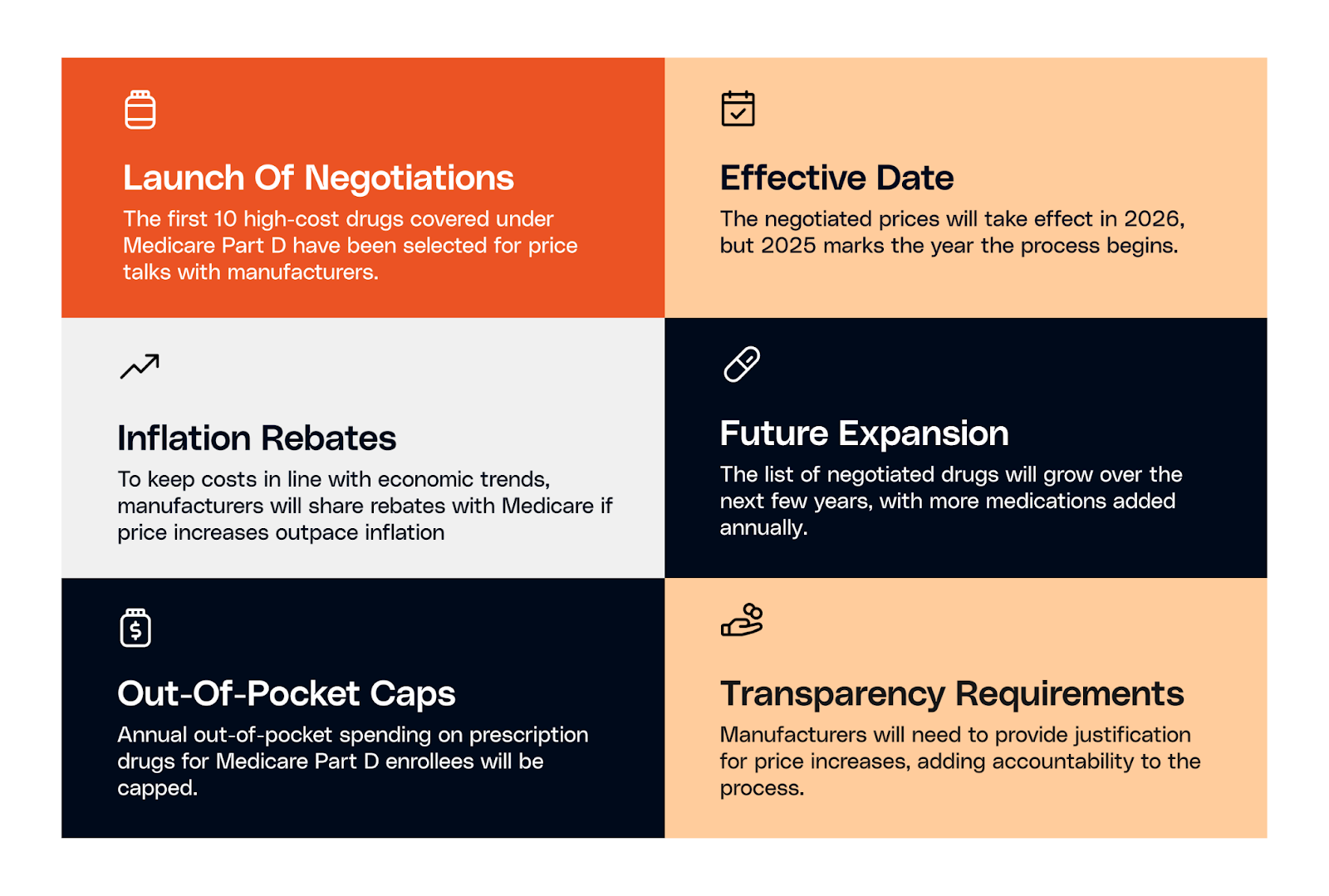
Now, with the next wave of drugs already identified, the program is moving into its next phase.
Medicare Drug Pricing Reform: Why It Matters
The impact of the CMS drug pricing reform on health systems and pharma companies is significant in the following ways:
- Greater predictability in partnerships with payers: With negotiated prices and inflation safeguards, the drug costs will become more stable. This helps insurers plan benefits but also gives manufacturers clearer expectations in their contracting and market strategies.
- Closer alignment with providers: Doctors and care teams see the difference when patients can stick to their prescriptions. This also improves outcomes. For pharma companies, this reinforces the value of their therapies and strengthens trust with communities.
- A stronger future for Medicare: Working directly with manufacturers helps Medicare ensure that the program stays financially sound and sustainable. This also helps pharma companies continue to reach millions of people who depend on their medications.
Impact on Pharma: What Companies Need to Know
Medicare Drug Price Negotiation Impact on Pharma
Medicare’s drug pricing negotiation program marks a major shift for the pharmaceutical industry. Once a drug is selected, manufacturers work directly with CMS to set prices, often leading to revenue projections that look very different from the past.
The impact won’t stop at Medicare. Private insurers are expected to follow these negotiated benchmarks, pushing drugmakers to revisit pricing strategies, forecasting models, and long-term R&D investments. The reform also raises expectations for transparency. Manufacturers must now justify price increases and adapt to new affordability pressures that may influence how therapies move through approval and launch.
What Manufacturers Should Do Now
For drugs that will be negotiated in 2026, preparation must begin well before formal discussions with CMS. Companies in negotiations in 2025 started building strategies months in advance, those approaching the next cycle can’t afford to wait.
Key priorities include:
1. Build negotiation strategies early by assessing the product’s clinical and economic impact and gathering real-world evidence to support pricing.
2. Model revenue implications by anticipating how negotiated prices may influence financial forecasts.
3. Strengthen payer and stakeholder engagement by leveraging data to demonstrate value and differentiate therapies.
4. Prepare for increased scrutiny by developing clear, evidence-based justifications for pricing decisions.
Preparing Your Market Access Strategy for 2026
With upcoming Medicare drug price negotiations in 2026, life sciences companies need to rethink how they approach market access. Pricing, payer expectations, and patient affordability are changing quickly, and traditional strategies may fall short.
This is where Humbi by Innovaccer can make a difference. By combining comprehensive Medicare and Medicaid data with actuarial intelligence, Humbi helps pharma companies:
- Assess the payer landscape: Map coverage trends across states, track formulary changes, and understand payer decision-making.
- Optimize pricing and contracting: Model the impact of negotiations, rebates, and inflation penalties to strengthen payer discussions.
- Profile patient and provider segments: Identify utilization patterns, prescribing behaviors, and access barriers to shape targeted strategies.
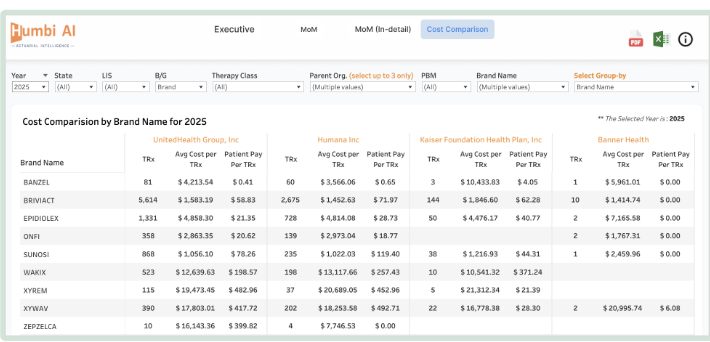
- Evaluate policy impact: Monitor regulatory changes, pricing pressures, and volume shifts in real time.
- Demonstrate value with evidence: Convert complex claims and outcomes data into compelling HEOR stories that resonate with payers.
In 2026, companies that rely only on traditional forecasts will be caught off guard. Those that leverage data-driven platforms can navigate uncertainty and protect margins while ensuring that patients get access to the therapies they need.
Preparing for a New Era in Drug Pricing
The reality is, 2025 was not the year where just another policy shift happened; it established significant changes in drug pricing, access, and reimbursement in the U.S. healthcare system.
Now, as 2026 approaches, the focus will shift from adjustment to optimization. Manufacturers, payers, and providers alike will need to operationalize lessons from the first negotiation cycle, refine data strategies, and anticipate broader ripple effects across commercial markets.
Those who treat this not as a compliance exercise but as a long-term shift in how value is defined through outcomes, equity, and evidence, will be the ones best positioned to navigate what comes next.
Ready to adapt your market access strategy for 2026? Discover how Humbi by Innovaccer can help you turn uncertainty into opportunity. Connect now.

